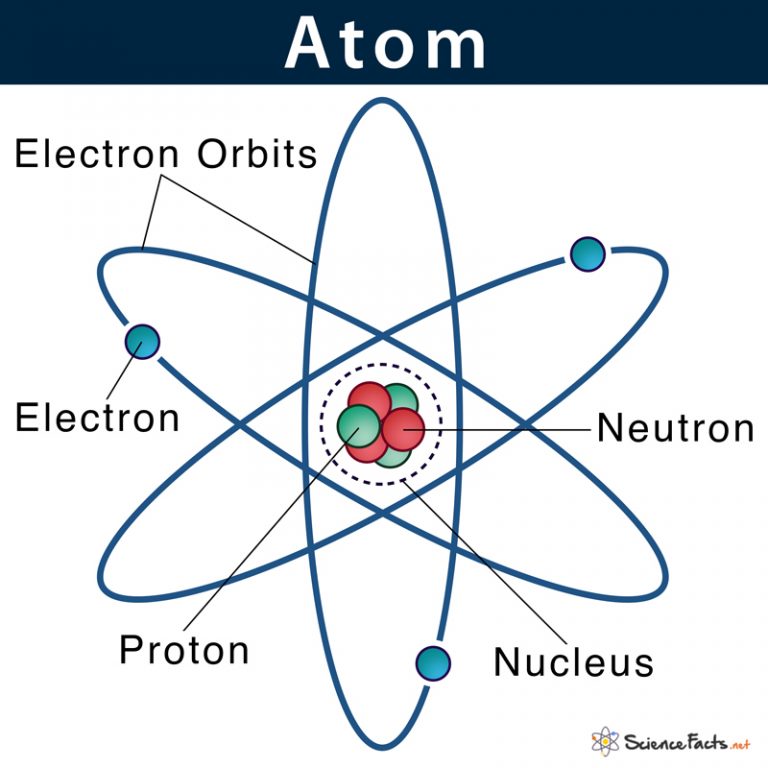
Atom Definition, Structure & Parts with Labeled Diagram
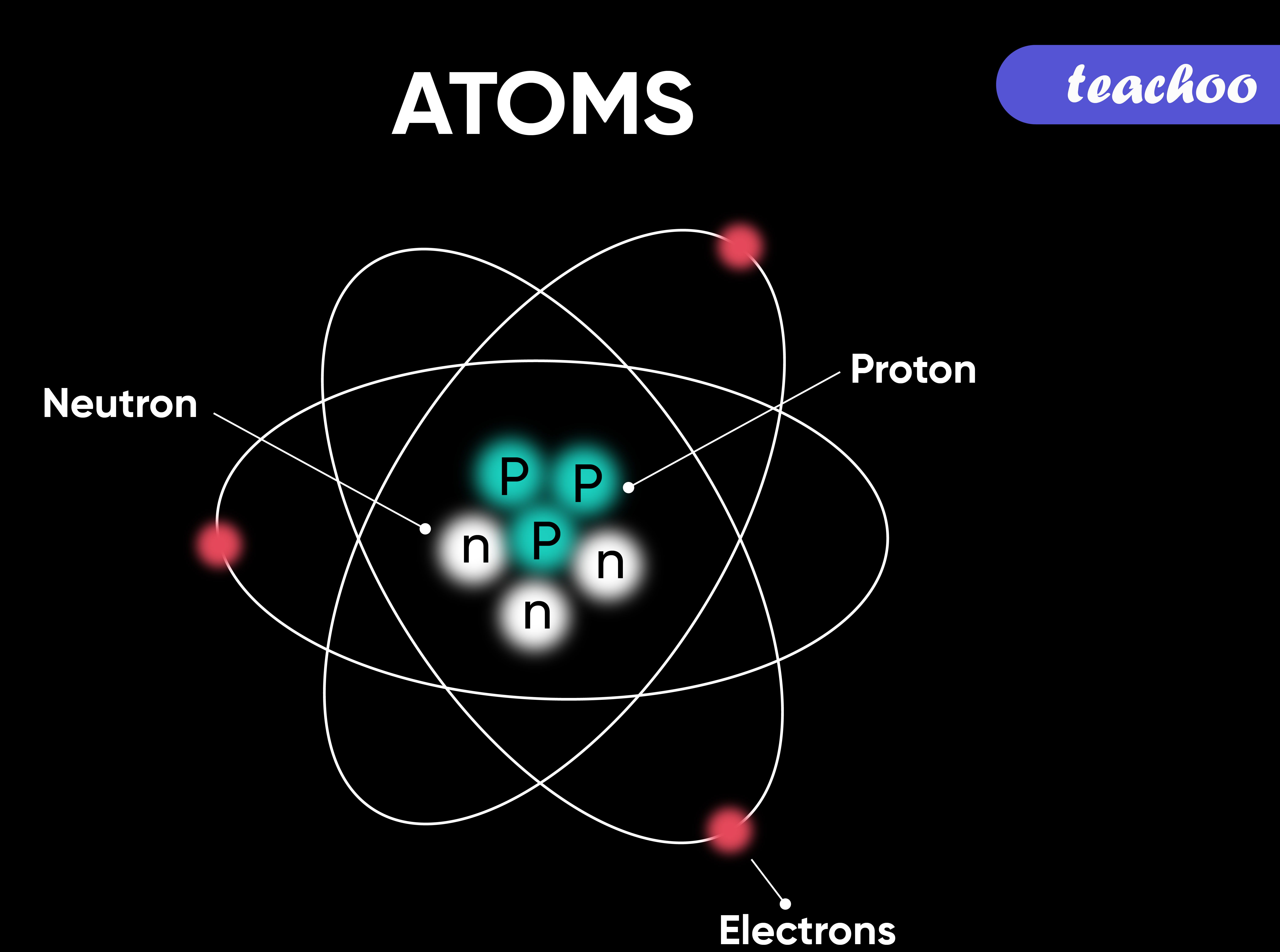
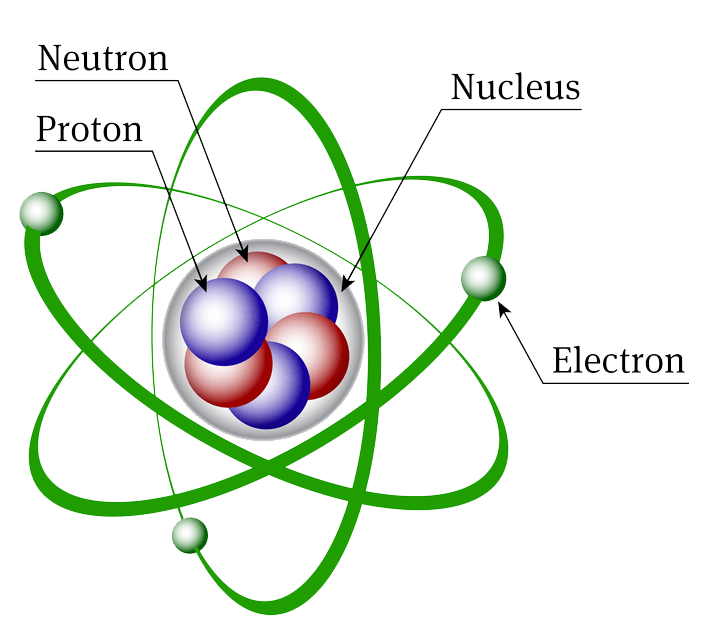
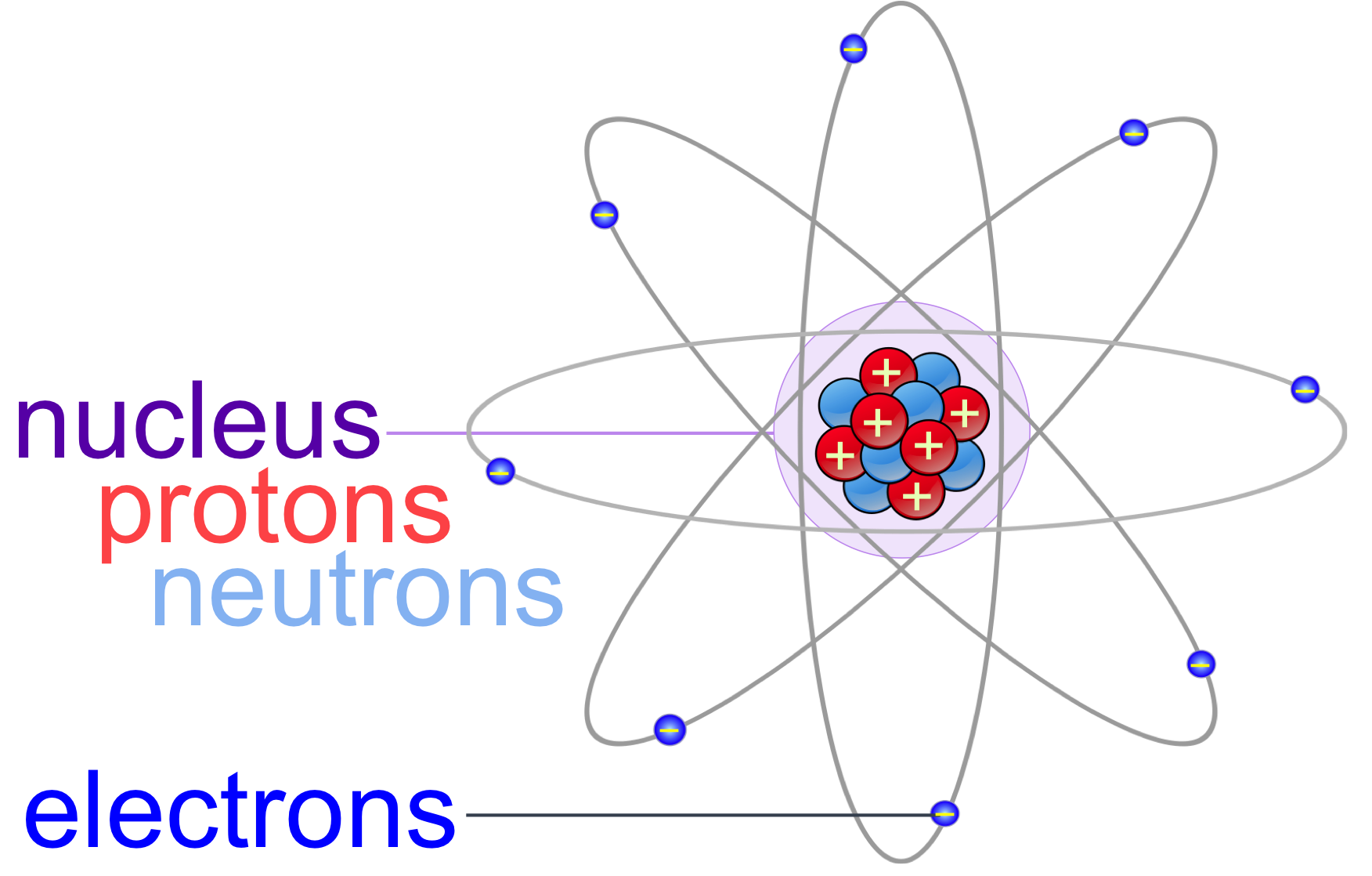
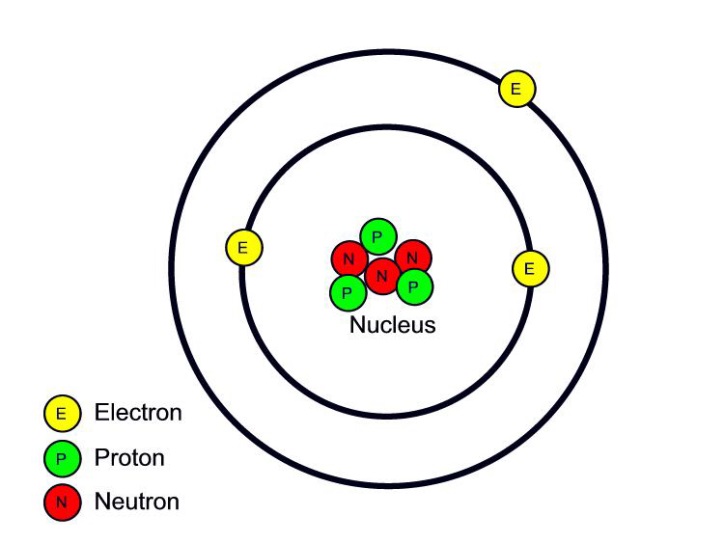
Atomic Structure All atoms except hydrogen contain three basic subatomic particles: 1) electrons, 2) protons, and neutrons. Neutrons and protons are found at the center of the atom within a dense region called the nucleus. In contrast, electrons are found outside the nucleus in a region called the electron cloud or electron shell. 1) Electrons
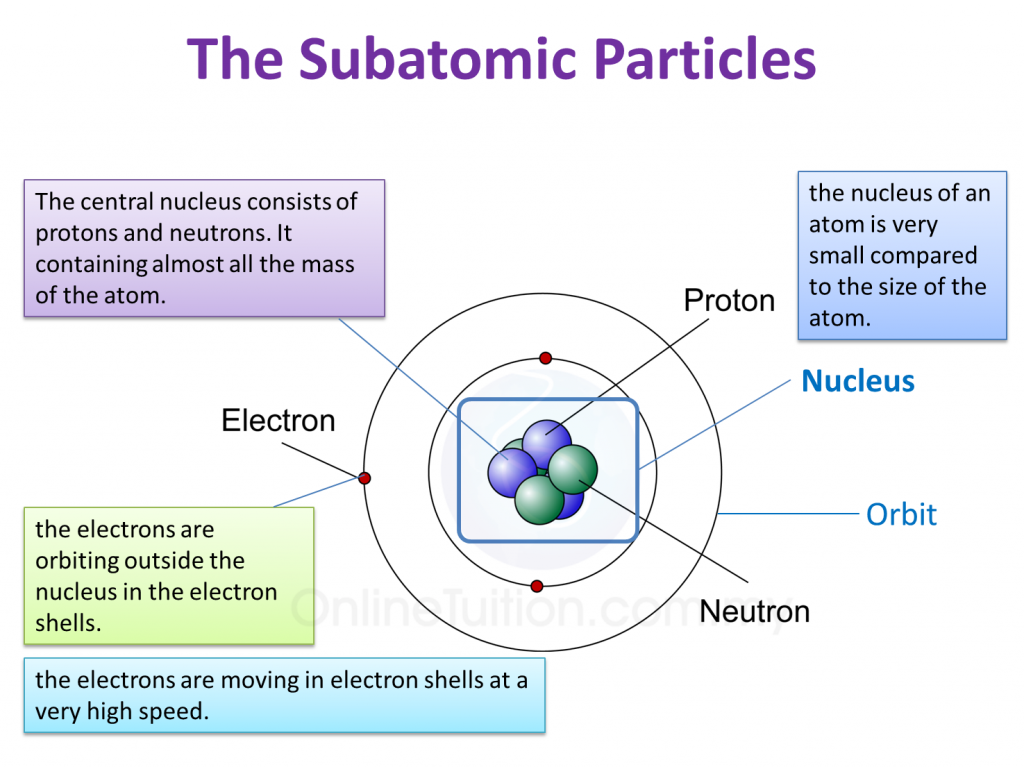
4.2 Structure of Atoms SPM Science
An atom is the smallest unit of matter that retains the chemical properties of an element. Atoms are able to interact with each other through bonding, to form more complex substances, also known as molecules. These interactions determine the state of matter the atoms are in, as they can be found as solids, liquids, or gases.

Atoms and Atomic Structure
Because of the definition of the unified atomic mass unit, each carbon-12 atom has an atomic mass of exactly 12 u, and so a mole of carbon-12 atoms weighs exactly 0.012 kg. Neils Bohr's model a.
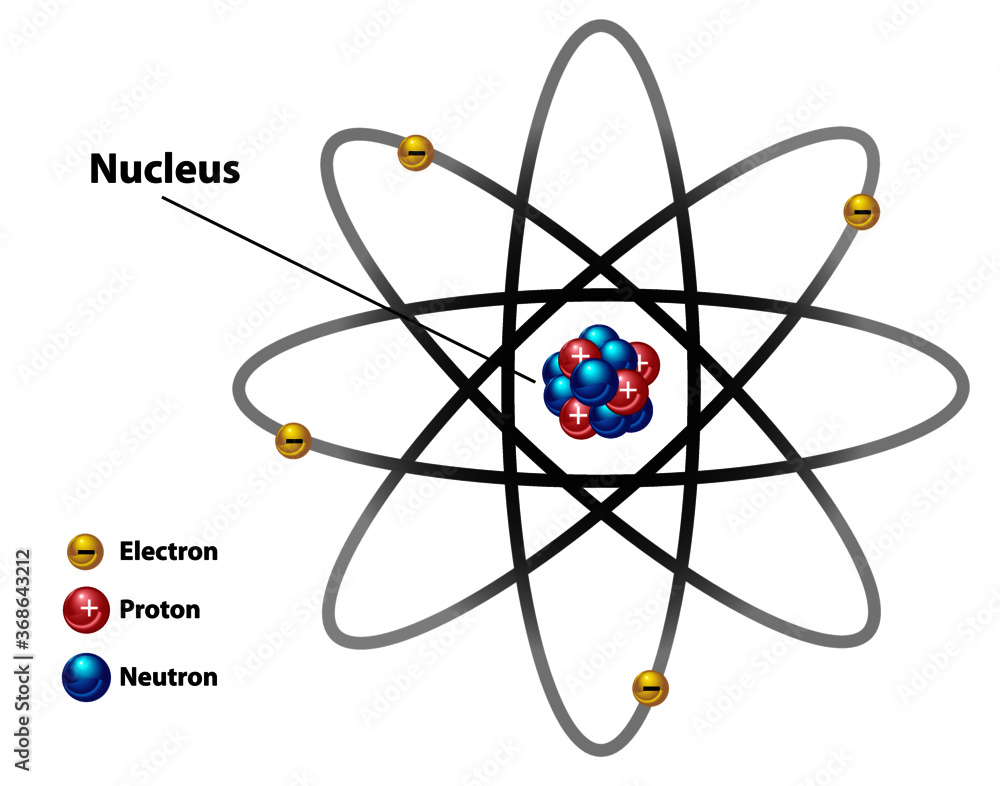
Atomic nucleus diagram labeled with electron, proton, and neutron. Stock Vector Adobe Stock
Table 1. Properties of Subatomic Particles Charge (C) Mass (amu) Mass (g) −1.602 × 10 0.00091 × 10 The number of protons in the nucleus of an atom is its atomic number (Z). This is the defining trait of an element: Its value determines the identity of the atom.
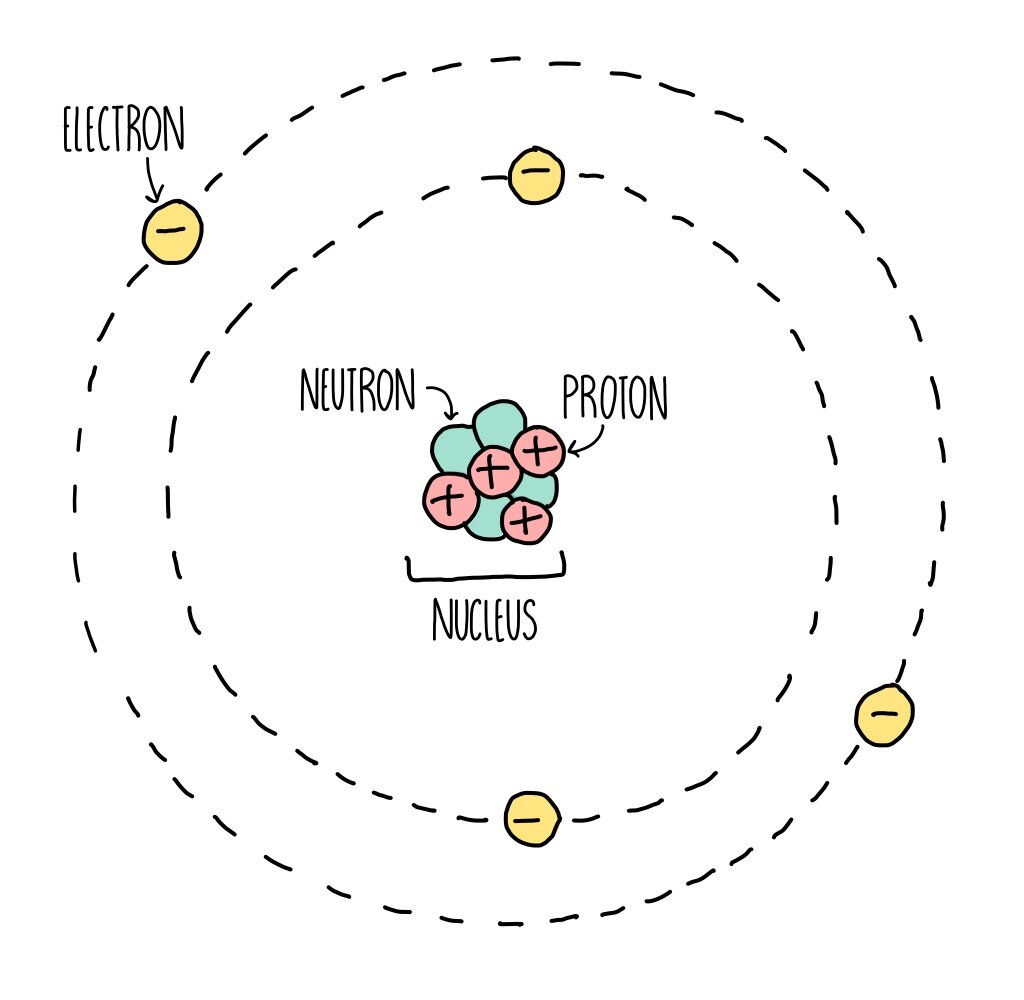
Atomic Structure (GCSE) — the science sauce
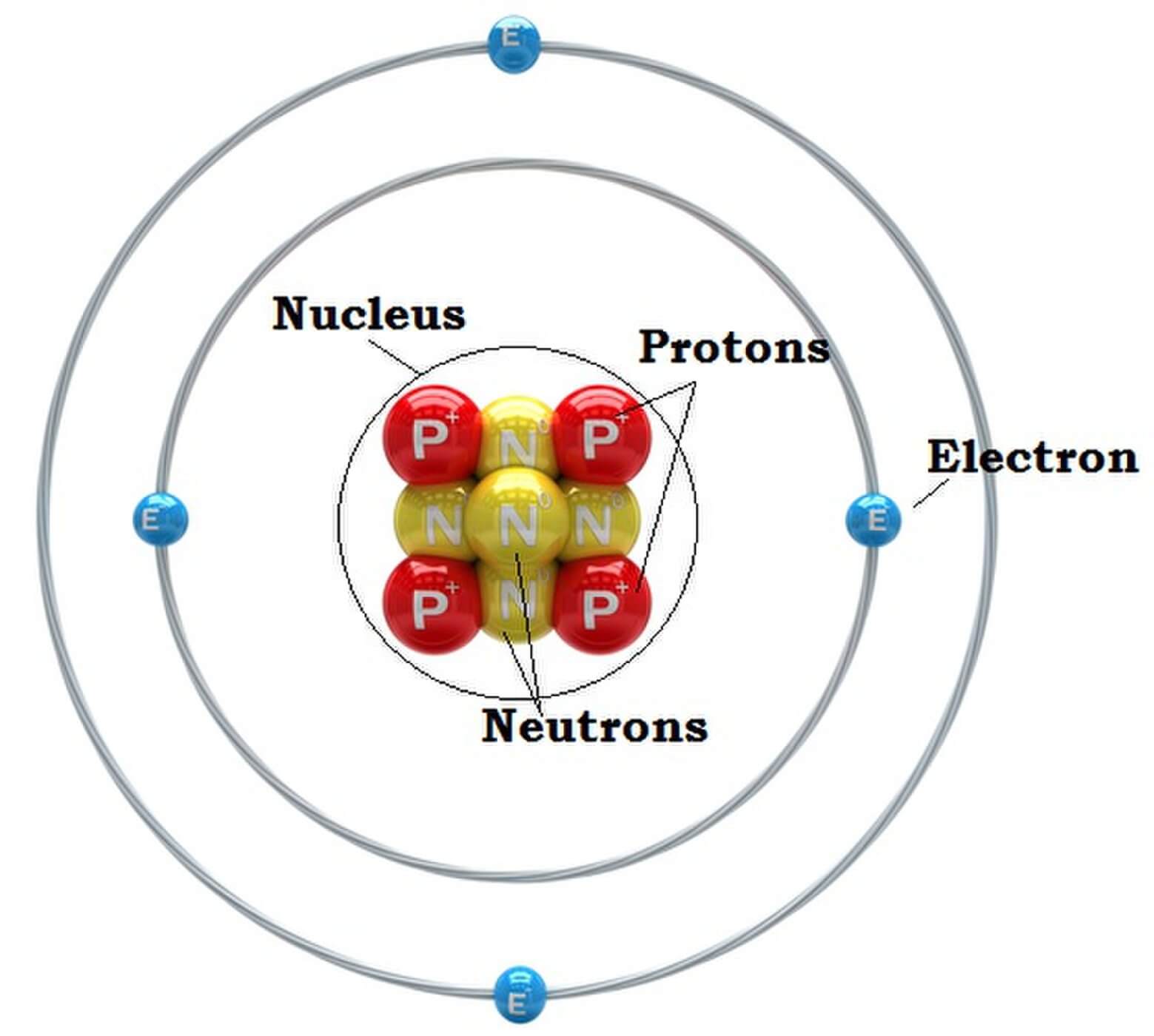
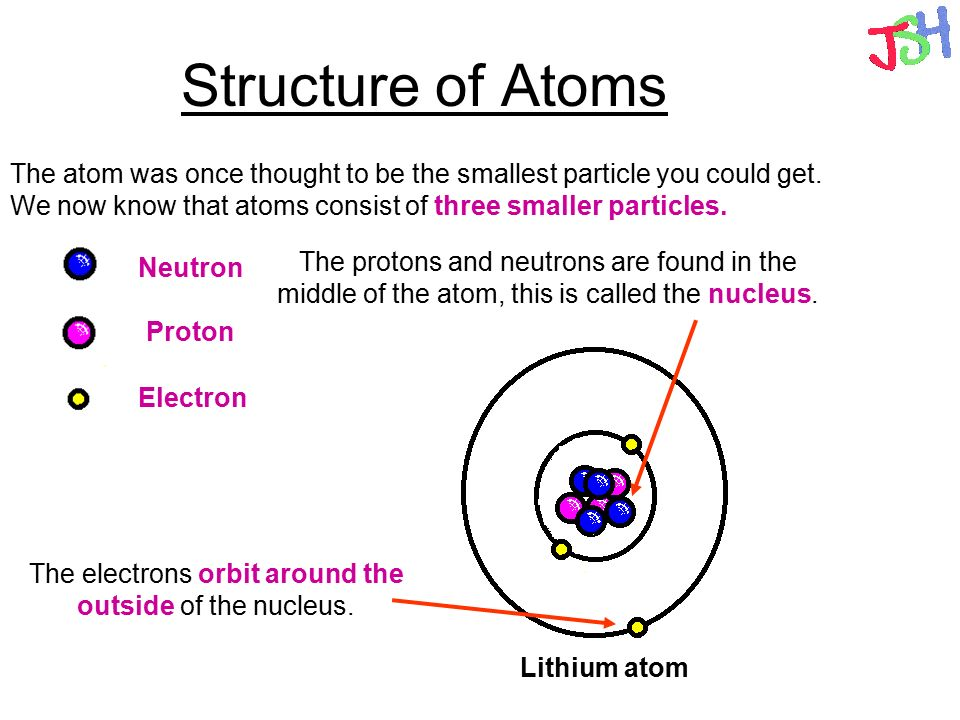
GCSE AQA Trilogy Atomic structure - AQA Structure of the atom Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles.
What is Atom? How does it Exist? and it's Symbols Teachoo
An atom that gains one or more electrons will exhibit a negative charge and is called an anion. Positively charged atoms called cations are formed when an atom loses one or more electrons. For example, a neutral sodium atom (Z = 11) has 11 electrons. If this atom loses one electron, it will become a cation with a 1+ charge (11 − 10 = 1+).
Structure of an Atom Structure & Use of Electron & Proton in Electronics
Atomic or ionic charge = number of protons − number of electrons. An atom that gains one or more electrons will exhibit a negative charge and is called an anion. Positively charged atoms called cations are formed when an atom loses one or more electrons. For example, a neutral sodium atom (Z = 11) has 11 electrons.
What is an Atom? Definitions & Examples Let us learn Basics News Bugz
The periodic table By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior. Devised by Russian chemist Dmitri Mendeleev (1834-1907) in 1869, the table places elements into columns— groups —and rows— periods —that share certain properties.
/GettyImages-141483984-56a133b65f9b58b7d0bcfdb1.jpg)
35 Label The Parts Of The Atom In The Diagram Below Labels For Your Ideas
Parts of an Atom An atom consists of two parts. These are the nucleus and extranuclear portions. The nucleus is present in the centre of the atom and is surrounded by the extranuclear portions. The radius of the nucleus of an atom is nearly 10 - 15 m, while that of the atom is 10 - 10 m.

Atomic Structure Broad Learnings
The electron configuration and orbital diagram of helium are: The n = 1 shell is completely filled in a helium atom. The next atom is the alkali metal lithium with an atomic number of 3. The first two electrons in lithium fill the 1 s orbital and have the same sets of four quantum numbers as the two electrons in helium.
Label Parts of an Atom — Learning in Hand with Tony Vincent
An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Protons and neutrons have approximately the same mass, about 1.67 × 10 -24 grams, which scientists define as one atomic mass unit (amu.
Atomic Structure Biochemistry
Chemical Symbols. A chemical symbol is an abbreviation that we use to indicate an element or an atom of an element. For example, the symbol for mercury is Hg (Figure \(\PageIndex{3}\)). We use the same symbol to indicate one atom of mercury (microscopic domain) or to label a container of many atoms of the element mercury (macroscopic domain).
The Structure of the Atom GCSE Physics Science) AQA Revision Study Rocket
Relative charge. -1. The number of electrons in an atom is always the same as the number of protons, so atoms are electrically. neutral. overall. Atoms can lose or gain electrons. When they do.
Atoms & Molecules echapter — The Biology Primer
All atoms are roughly the same size, whether they have 3 or 90 electrons. Approximately 50 million atoms of solid matter lined up in a row would measure 1 cm (0.4 inch). A convenient unit of length for measuring atomic sizes is the angstrom (Å), defined as 10 −10 metre. The radius of an atom measures 1-2 Å.

Structure of an atom universalxoler
The orbits are labeled by an integer, the quantum number n. Electrons can jump from one orbit to another by emitting or absorbing energy. The inset shows an electron jumping from orbit n=3 to orbit n=2, emitting a photon of red light with an energy of 1.89 eV. (more)
Atomic structure WGHS Junior Science
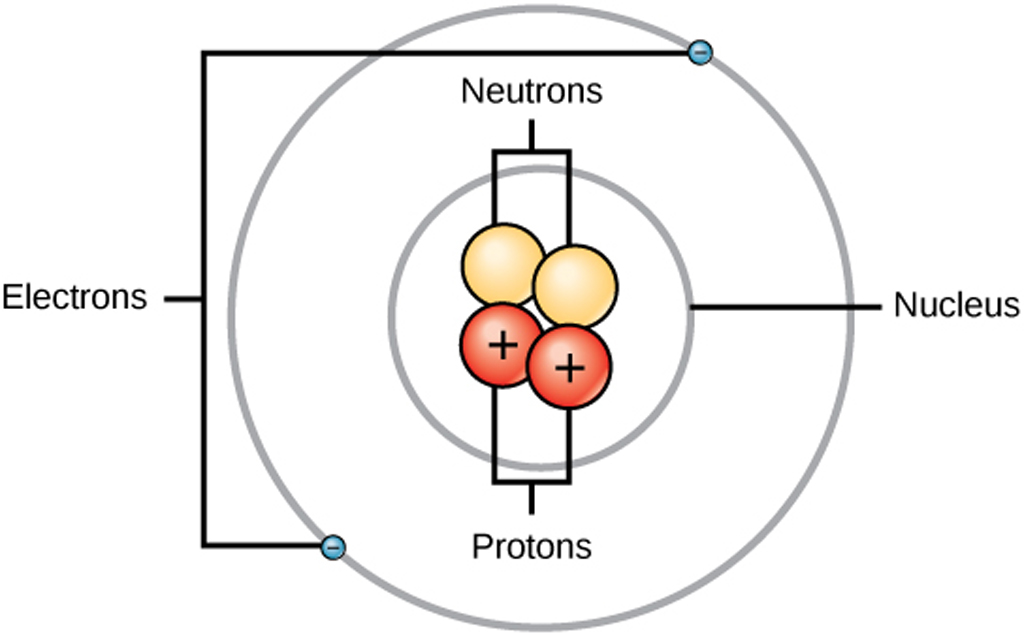
Figure 2.2.1 2.2. 1: The Structure of the Atom. Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different.